An overview of Alzheimer’s disease: what are the risk and protective factors, signs and symptoms, diagnosis, assessment, causes and screening.
SUMMARY
- The number of cases increases with age
- Factors that increase the risk
- Protective factors
- Signs and symptoms
- The signs that should worry us?
- Diagnostic
- Assessment
- Etiology
- Screening
Alzheimer’s disease is the most common dementia with 60% of dementia cases.
- 95% of cases are sporadic, affecting individuals in an isolated and irregular manner at a late age (over 65).
- Hereditary forms (which are characterized by the transmission of ‘bad genes’) represent only about 5% of total cases. The genes involved in the family forms are the presenilin 1 and 2 genes (PSEN), leading to the onset of the disease in middle age (around 30 to 40 years depending on the mutated gene). The mutation being dominant, and since each parent transmits an allele (variant of the gene), a child is 50% likely to develop the disease.
There are also genetic predispositions caused by the mutation of certain genes, in particular the apolipoprotein E gene (APP) which increases the risk of developing the disease. The mutation is not dominant.

The number of Alzheimer’s disease cases increases with age
Five percent (5%) of people aged 65 and over are affected by the disease (8% suffer from dementia) in industrialized countries. The prevalence rate (total number of cases) increases exponentially with age, approximately doubling every 5 years. Fifteen percent of patients are under 75 years old. According to a report published in 2012 by the World Health Organization, Alzheimer’s disease affects from the age of 65 2.3% of men and 3% of women and affects 33% of men over 90 and 48. % of women of the same age. The table below summarizes the prevalence rate by age group and according to sex, which demonstrates that age is therefore the main risk factor and that women are more affected than men.
The tables below summarize incidence (number of new cases) and prevalence by age and gender.
| Age | Prevalence rate | Impact |
| Men | ||
| 65-69 years old | 1 | 0.6 |
| 70-74 years old | 3 | 1.5 |
| 75-79 years old | 6 | 1.8 |
| 80-84 years old | 12 | 6.3 |
| >85 years old | 20 | 8.8 |
| Women | ||
| 65-69 years old | 1 | 0.7 |
| 70-74 years old | 3 | 2.3 |
| 75-79 years old | 6 | 4.3 |
| 80-84 years old | 14 | 8.4 |
| >85 years old | 31 | 14.2 |
Sources: EUROMED, Paquid (France) The table below summarizes the incidence (number of new cases) according to age and sex.
Factors that increase the risk of Alzheimer’s disease
- Age (main risk factor).
- The low level of study.
- The female gender.
- Family history.
- History of head trauma.
- Depression.
- Diabetes.
- Untreated hypertension.
- Genetic factors: in the sporadic forms of Alzheimer’s disease, a susceptibility factor has been demonstrated (the Epsilon 4 form of apolipoprotein E).
- Toxic substances (controversial hypothesis at present).
Protective factors
- Chronic use of nonsteroidal anti-inflammatory drugs. (hypothesis to be confirmed)
- A healthy diet (Mediterranean diet).
- Physical activity.
- Cognitive activity.

The multiplicity of these factors (probably interdependent) would explain why this disease presents itself under very diverse aspects and affects people unequally: thus it can affect a person aged 40 or 80 and its average duration after diagnosis varies from 5 to 8 years.
The signs and symptoms

day-to-day memory.
The stages of Alzheimer’s disease
There are different stages of Alzheimer’s disease depending on the severity of the symptoms:
Moderate stage
The disease is characterized by the insidious appearance of memory disorders (often denied by the patient) which evolve very gradually. These disorders may be accompanied by depression and/or anxiety. Memory problems gradually worsen, and other cognitive functions (e.g. planning, calculation) begin to be affected, leading to a (partial) inability to carry out certain activities of daily living (phoning, taking the bus, managing one’s budget).
Moderate to severe stage
The patient suffers from the advanced stage of anosognosia (he is no longer aware of his disorders), is disoriented in space and time. He has trouble communicating with those around him because of language, gnosis, praxis and attention disorders. The loss of autonomy now affects the basic activities of daily life (dressing, personal hygiene, moving around, toilets). Behavioral disturbances can occur at this stage and include agitation, hallucinations, aggressiveness towards those around them, delusional manifestations, sleep disturbances, apathy, ambulation, vocalization and stereotyped behaviors.
Severe stage
The patient has completely lost his autonomy. He eats less and less, weakens, facilitating the appearance of other mental disorders such as confusion, depression or slip syndrome (the patient lets himself die by refusing to eat). Other complications appear: falls with fractures, bedsores… The death of the patient occurs between 5 and 10 after the date of the first symptoms. The profile of clinical manifestations is given as an indication, as they are heterogeneous. In some patients, the symptoms start suddenly, while in others, the diagnosis of dementia can be made, for example, following a surgical operation (an operation sometimes causes mental confusion in an elderly person.
Diagnostic
in 1984, a group of researchers from NINCDS-ADRDA (National Institute of Neurological and Communicative Disorders and Stroke and Alzheimer’s Disease and Related Disorders Association) established diagnostic criteria. There are three diagnostic levels: possible (pre-mortem diagnosis), probable (pre-mortem diagnosis) and definitive (post-mortem diagnosis after biopsy).
These criteria are:
– Cognitive disorders (memory, language, judgment, reasoning, etc.) generally accompanied by behavioral and psychological disorders (agitation, depression, aggressiveness, wandering, insomnia, etc.) that can affect activities of daily living.
The symptoms of dementia have two main characteristics:
- Symptoms appear in adulthood (mental retardation is not one of them).
- The symptoms are in most cases progressive and irreversible (confusional and depressive states of the elderly are not included).
In 2010, the American Institutes of Health (National Institute of Health) adopted new diagnostic criteria for Alzheimer’s disease. They now take into account the breakthroughs made in neuroscience that have allowed a better understanding of the disease.
While the clinical criteria used since then described the last stage of disease progression (when symptoms of dementia had already manifested), the new guidelines consider all known stages of disease development, which progresses gradually and can cause brain changes over a decade before it becomes apparent.
The new criteria include three phases:
1. The first so-called preclinical phase. She describes changes in the brain, including the formation and accumulation of senile plaques made up of beta-amyloid aggregates. These plaques would be responsible for the death of neurons. The use of brain neuroimaging techniques and biomarkers* is now helping to determine if changes in the brain are warning signs of disease.
2. The second phase. It corresponds to episodes of memory loss that are sufficiently obvious to be measured and noted by those around them. On the other hand, they are not pronounced enough to cause the loss of autonomy.
3. The third and final phase. This is the phase of dementia currently described by the medical community.
* Neurologists are increasingly using biomarkers to observe the evolution of pathology. However, they cannot be routinely used to make a clinical diagnosis.
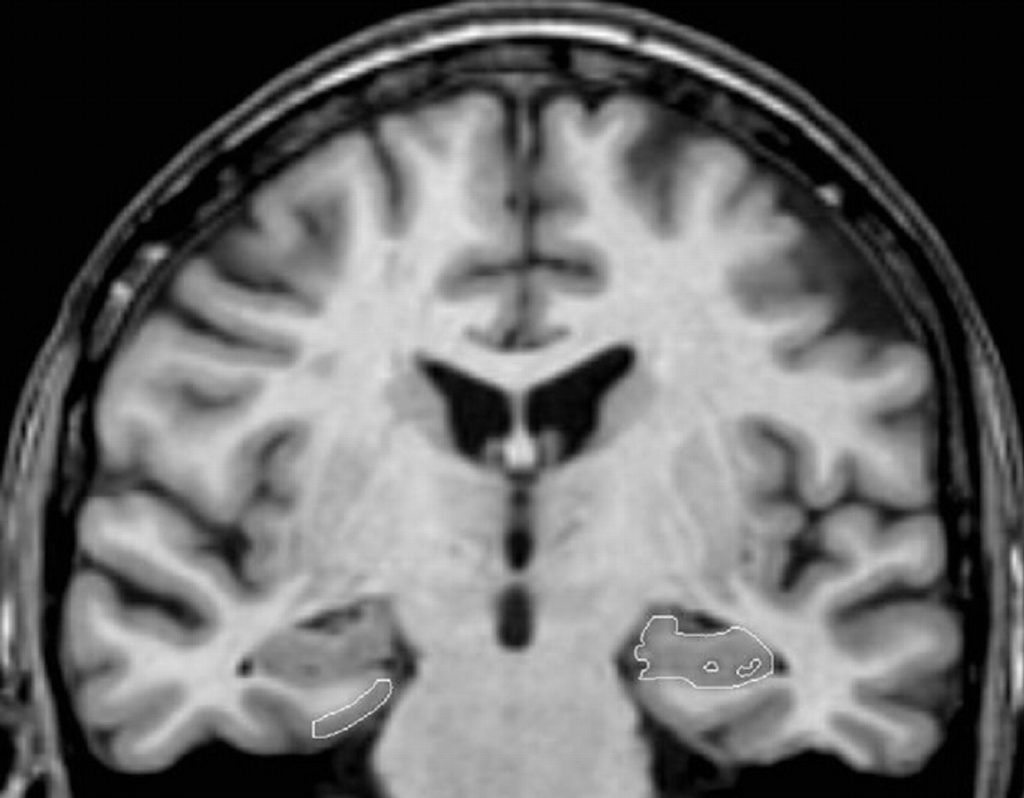
Complementary examinations (in particular neuroimaging examinations) do not contribute to the diagnosis of Alzheimer’s disease. They most often indicate enlargement of the cerebral ventricles and widening of the cortical sulci, reflecting neuronal loss.
However, this aspect can be observed in healthy elderly people, as well as in patients suffering from other forms of dementia. However, hippocampal atrophy is considered specific for Alzheimer’s disease, and may aid in diagnosis.
According to the American psychiatric manual DSM-IV, the patient must fulfill criteria encompassing the following symptoms:
A. Appearance of multiple cognitive deficits as evidenced by both:
1. memory impairment;
2. one or more of the following cognitive disturbances:
- Aphasia (language disturbance);
- apraxia (impaired ability to perform an activity despite intact motor functions);
- agnosia (inability to recognize objects despite intact sensory functions);
- disturbances of executive functions (making plans, organizing in time, having an abstract thought).
B. Cognitive deficits in criteria A1 and A2 represent a significant decline from previous level of functioning.
C. The course is characterized by a gradual onset and continuous cognitive decline.
D. The cognitive deficits of criteria A1 and A2 are not due to:
- other conditions of the central nervous system (eg cerebrovascular disease, Parkinson’s disease, brain tumour);
- general conditions that can lead to dementia (eg hypothyroidism, vitamin B12 deficiency, HIV infection, etc.) substance-induced conditions.
E. Deficits do not occur exclusively during delirium.
F. The disturbance is not better accounted for by a disorder such as depression or schizophrenia.
Assessment
The evaluation of Alzheimer’s disease includes an interview with the patient and a family member, neurological tests, clinical examinations and neuroimaging.
Neuroimaging techniques can confirm or invalidate the diagnosis.
These techniques are:
• The electroencephalogram (EEG): this examination aims to see the electrical activity of the brain.
• Single photon emission tomography (SPECT): this assesses blood flow and the volume of cerebral regions.
• Positron emission tomography (PET): this studies the activity of brain regions. For example, patients with Alzheimer’s disease have reduced activity in the temporal and parietal lobes.
• Computed tomography: this measures the thickness of the regions of the brain affected in Alzheimer’s disease.
• Magnetic resonance imaging ( MRI): this visualizes changes in the structure of the brain.
• Functional magnetic resonance imaging (fMRI): this measures the functional activity of brain regions.
Blood and urine and cerebrospinal fluid tests to reveal vitamin deficiency, hormonal dysfunction, the presence of toxic agents or molecules involved in Alzheimer’s disease (e.g. amyloid protein)
Medical history ( e.g. history of head trauma, use of substances that may alter mental status).
Neuropsychological tests: the neuropsychological assessment contributes to the diagnosis in mild to moderate forms, by emphasizing the importance of memory disorders in relation to the deficit of other cognitive functions.
The MMSE is the most frequently used test because it quickly gives valuable information about short-term memory and other cognitive functions.
The other tests are the Clinical dementia Rating Scale, the ADAS scale (Alzheimer’s Disease Assessment Scale), the clock test, the free and cued recall test according to the procedure of Grober and Buschke and the five-word test.
Alzheimer’s disease, in the early stages, can be diagnosed through two neuropsychological examinations carried out +/- 6 months apart.
This delay will inform the doctor about the progressive nature of the disease (the evolution confirms the initial diagnosis).
Behavioral tests ( CERAD Behavior Rating Scale for Dementia, the neuropsychiatric inventory) are used to assess the frequency and severity of behavioral disorders.
Functional tests assess the degree of autonomy of patients, for example instrumental activities of daily living.
The causes
The causes of the disease remain unknown, but it is already strongly suspected that neuronal death is due to the abnormal accumulation of a protein called amyloid in the brain of Alzheimer’s patients, resulting in the form of aggregates in the form of amyloid plaques. around neurons.
Accumulating amyloid causes neuroinflammation, characterized by the production of cytokines, and oxidative stress, characterized by the production of free radicals.
These two phenomena would in turn increase the formation of plaques.
On the other hand, Alzheimer’s disease is characterized by neurofibrillary degeneration (NFD) which is characterized by lesions affecting the interior of neurons.
These DNFs are formed from an accumulation of a protein called tau protein which is present in an abnormal form.
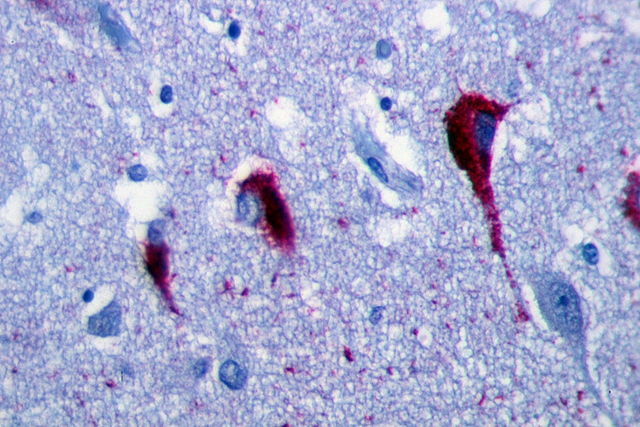
These lesions are distributed throughout the brain, but predominate in the hippocampus and entorhinal cortex, a brain structure involved in memory storage.
Amyloid accumulation can occur 10 to 15 years before the first symptoms of Alzheimer’s disease, while tau protein begins to accumulate in the neocortex 3 to 5 years before the first symptoms.
Changes in amyloid and tau levels can be detected in cerebrospinal fluid, as well as by neuroimaging techniques (e.g. imaging using an amyloid-scoring probe).
The disease is also characterized by neuronal loss at the cortical level and a marked drop in acetylcholine, the neurotransmitter involved in memory.
Screening
Extensive efforts have been made to identify biomarkers in cerebrospinal fluid and blood. The measurement of the ratio between the tau protein and the amyloid protein in the blood is a reliable indicator of a pathology in subjects suffering from Alzheimer’s disease, but also in those who have not yet developed the disease.
Neuroimaging (e.g., positron emission tomography, MRI) is used to monitor disease progression, determine severity, and measure the effectiveness of treatment.
The use of a probe marking amyloid makes it possible to follow the accumulation of this protein and to identify the individuals who will develop the disease years later.
