Lewy body dementia is characterized by cognitive decline, visual and spatial disturbances, fluctuations in alertness and attention, hallucinations and motor disturbances.
Most cases of Lewy body dementia are sporadic. Some hereditary cases have been reported.
Historical
1912: the scientist Friederich Lewy observes for the first time ‘inclusions’ of about ten microns (one thousandth of a millimeter) in diameter, inside neurons. These inclusions, made up of deposits of proteins called alpha-synuclein, had previously been described in Parkinson’s disease. These inclusions are subsequently called Lewy bodies.
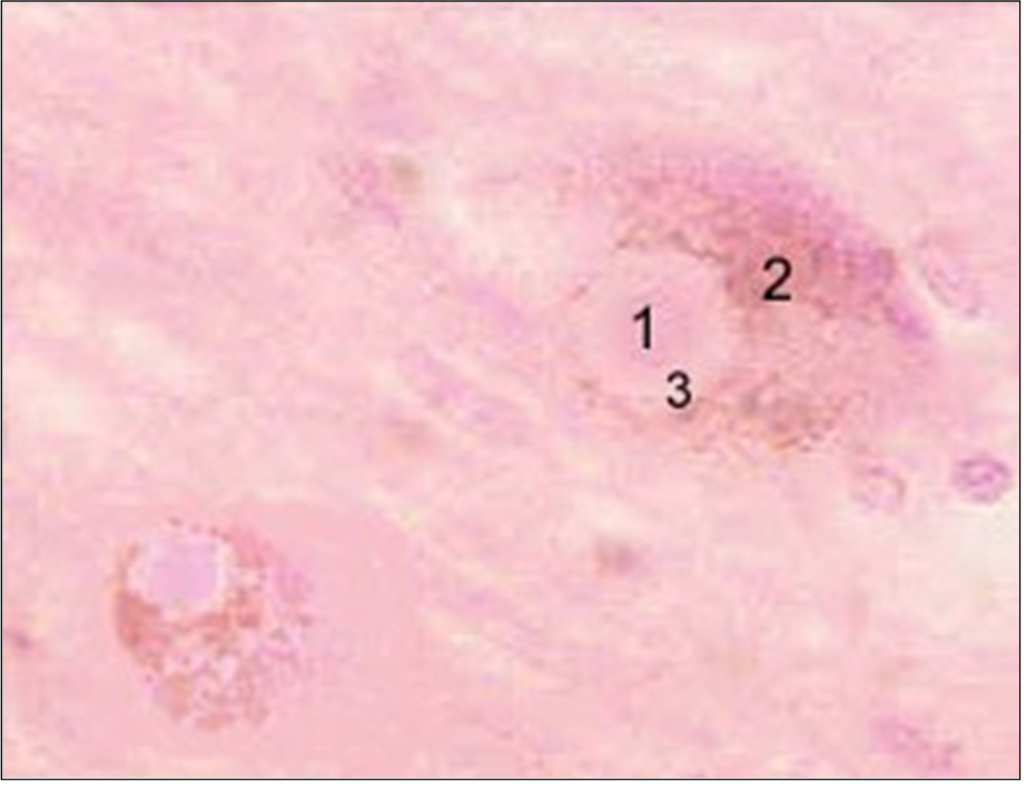
1923: Friederich Lewy reports the existence of mental disorders in patients with Parkinson’s disease. He suggests the presence of ‘foreign bodies’ (later called Lewy bodies by Prof. Tretiakoff) inside neurons.
1961: Professor Okazaki reports the case of two patients with dementia associated with behavioral and motor disorders. The autopsy reveals the presence of Lewy bodies in the cerebral cortex.
1984: Pr Kosaka proposes the term Lewy body disease after collecting several cases of dementia patients. These patients presented with Lewy bodies in the brainstem and in certain regions of the temporal lobe related to the limbic system (anterior cingulate cortex, amygdala, and hippocampus).
1996: A group of researchers hold a conference to define diagnostic criteria for what is now called Lewy body dementia (abbreviated as LDB). LDB is therefore defined as a neurodegenerative dementia grouping the following terms: diffuse Lewy body disease, cortical Lewy body disease, Lewy body disease, senile dementia of the Lewy type, dementia with Lewy bodies, the variant with Lewy bodies of Alzheimer’s disease.
This working group revised the clinical and pathological diagnostic criteria and included new recommendations on how to better detect clinical symptoms.
Epidemiology
LDB accounts for 10% to 22% of post-autopsy dementia cases. By way of comparison, Alzheimer’s disease and vascular dementia account for 40-70% and 10-15% of dementia cases respectively. It is therefore the second leading cause of dementia.
The average age of onset is between 50 and 70 years, and affects more particularly men. A minority of MCI patients also have Parkinson’s disease. However, unlike patients with Parkinson’s, motor symptoms are usually mild and not accompanied by resting tremors. Most people with MCI have symptoms and lesions characteristic of Alzheimer’s disease.
Genetic factors
Most cases of LDB are sporadic, that is, affecting individuals irregularly. However, there are genetic mutations that can increase the risk of developing a sporadic form of the disease.
There are also family forms of the disease, explained by the mutation of genes (for example, the gene for alpha-synuclein).
Histological lesions of Lewy body dementia
Lewy bodies. These typical lesions contain proteins called alpha-synuclein and ubiquitin. These Lewy bodies are found inside neurons located in cortical (hippocampus, cingulate cortex, amygdala, basal nucleus of Meynert, hypothalamus) and subcortical (substance nigra, locus coeruleus, raphe nucleus, motor nucleus of vagus nerve) of the brain. The regions of the frontal cortex are less affected.
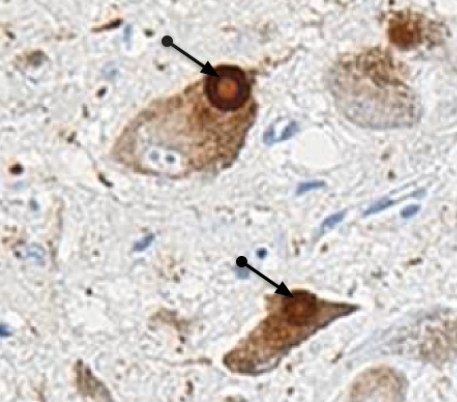
The presence of Lewy bodies in cortical regions seems to be related to the deterioration of intellectual functions.
Extensions of Lewy bodies are observed in the regions of the limbic system and the temporal lobe.
Lewy bodies and Alzheimer’s disease. A notable proportion of LDB patients present with lesions suggestive of Alzheimer’s disease: deposits of paired filaments in a helix (also called neurofibrillary tangles) and amyloid plaques. However, these filament deposits are less common in the brains of patients. According to the Braak scale, their number is between those of a healthy subject of the same age and that of an Alzheimer patient. On the other hand, the number of amyloid plaques is substantially identical in the two diseases, and appear more diffuse in LDB.
Lewy bodies and Parkinson’s disease. Lewy bodies are detected in the subcortical regions (substantia nigra and locus coeruleus) of brains of Parkinsonian patients.
Neuroimaging shows mild hippocampal atrophy in LDB compared to brains of healthy subjects. The activity of the primary visual cortex is reduced in patients with MCI.
Other types of lesions – called spongioses – appear in regions of the temporal lobe, particularly the tonsil.
Changes in neurotransmitters
The presence of these lesions in certain specific regions of the brain obviously has repercussions on the functioning of neurons.
Dementia with Lewy bodies is accompanied by a loss of neurons in the substantia nigra and the basal nucleus of Meynert, resulting in a decrease in the production of neurotransmitters called dopamine (DA) and acetylcholine (Ach), respectively.
Ach is the neurotransmitter of memory and attention, while DA is involved in motor control.
The death of neurons secreting Ach is probably responsible for cognitive disorders affecting in particular attention, concentration, visuo-spatial abilities, as well as visual hallucinations.
Motor disorders of the parkinsonian type are caused by a loss of neurons in the substantia nigra producing AD. Neuronal loss has also been described in the locus coeruleus, a region of the brain closely linked to the limbic system and synthesizing norepinephrine, an excitatory neurotransmitter. This neuronal death may be the cause of the behavioral disorders of paradoxical sleep observed in the disease.
Three main neural systems are therefore affected in LDB cholinergic (Ach), dopaminergic (DA) and noradrenergic (NA) systems.
The cholinergic system . Meynert’s basal nucleus contains neuron cell bodies (black circle) projecting to much of the cortex and the amygdala. These neurons synthesize acetylcholine, a neurotransmitter involved in memory storage and recall, motivation and attention. There is a significant loss of neurons from the basal nucleus of Meynert in DLB and Alzheimer’s disease (about two-thirds loss). The other acetylcholine-secreting nucleus is the septum (blue circle), whose neurons project into the hippocampus (blue arrow).
The dopaminergic system . The substantia nigra is made up of neurons (green circle) projecting into the striatum (a brain region involved in motor functions). These neurons synthesize dopamine and are particularly affected in DLB and Parkinson’s disease.
The noradrenergic system . The locus coeruleus (red circle), located in the brainstem, contains neurons projecting to the cortex, cerebellum and spinal cord (red arrows). These neurons synthesize norepinephrine, an excitatory neurotransmitter.
Clinical signs of Lewy body dementia
Clinical signs are characterized by both neurological, psychological and behavioral disorders, occurring insidiously and progressing over a period of several months to several years. The age of onset of LDB varies between 50 and 80 years. Men are slightly more affected than women.
According to the recommendations of the group of international experts, the essential criteria for the diagnosis of LDB are:
- Progressive dementia, i.e. progressive cognitive decline severe enough to interfere with activities of daily living (domestic and social activities).
- Predominance of visual and spatial disturbances (e.g. the patient no longer recognizes a route that he usually takes). Executive functions (reasoning, decision-making, abstract thinking, etc.) also decline. Visuospatial deficits are due to the presence of Lewy bodies in the visual associative cortex, located at the base of the temporal and occipital lobes. It should be noted that these lesions are not responsible for the slowing down of motor functions. The lack of perception is apparently at the origin of delusional disorders and misidentification (for example paramnesia, Capgras syndrome or the fact of not recognizing a relative). The patient also suffers from a significant attention deficit.
- Memory problems (in particular verbal memory) which do not necessarily appear at the start of the disease, but which gradually worsen.
In addition to these main criteria, there are so-called secondary criteria. Two criteria are sufficient for the diagnosis of probable MCI, one criterion for the diagnosis of possible MCI).
- Fluctuations in alertness and attention . These cognitive fluctuations are more marked and more frequent than in Alzheimer’s patients with a comparable cognitive deficit. They vary in duration and are similar to delusional disorders – they are sometimes called ‘pseudodelirium’. The characteristic signs are: loss of attention, incoherent language, hypersomnolence, impaired consciousness, staring into space. The patient suddenly returns to normal, confusing the family. The prevalence of fluctuations varies from 25 to 75%. Inexperienced physicians may have difficulty determining whether these fluctuations are secondary criteria for LDB.
A four-item questionnaire assesses the presence of these fluctuations with some precision. These items are:
- the person is sleepy and has a lethargic behavior during the day, although he sleeps through the night,
- she sleeps at least two hours during the day (before 7 p.m.),
- she has a lost look for long moments and
- sometimes disordered and confused thoughts.
This questionnaire detects with a high probability (positive predictive value* = 83%) the presence of DCL (and the absence of Alzheimer’s disease) if three of the four criteria are met.
Positive predictive value of a sign for a diagnosis: probability that the diagnosis is true if the sign is present.
- Visual hallucinations recurrent and detailed, sometimes accompanied by auditory hallucinations. They are one of the cardinal signs of the disease, although they also appear in other forms of mental illness (Alzheimer’s disease, vascular dementia, hallucinatory psychosis). Hallucinations occur early in the disease and are persistent. They come in the form of innocuous three-dimensional images, representing people, animals or objects. Patients suffering from visual hallucinations early generally see their physical and mental condition worsen more quickly. A deficit of acetylcholine in the temporal lobe is probably responsible for these visual hallucinations. It should be noted that the associative visual cortex of the temporal lobe contains Lewy bodies.
- Parkinsonian-like motor disorders. The vast majority of patients (up to 70%) show parkinsonian signs. These disorders are usually mild. They precede or follow the cognitive and behavioral disorders in a time interval not exceeding one year. If this is not the case, the doctor will make a differential diagnosis: other parkinsonian syndrome, dementia associated with Parkinson’s disease. Bradykinesia (slowness of body movements) is the main symptom. Gait disturbance, unsteady gait, limb stiffness and dystonia may occur as the disease progresses, or in response to treatment with antipsychotics (e.g. clozapine, risperidone) for soothe hallucinations. On the other hand, tremors at rest are rare.
Other criteria support the presence of a DLB:
- Orthostatic arterial hypotension and postural instability, resulting in dizziness, fainting, transient loss of consciousness, with or without falls. Orthostatic hypotension with syncope is observed in a quarter of patients.
- Hypersensitivity to antipsychotics (neuroleptics). Adverse effects suffered by patients with Lewy body dementia and treated with antipsychotics are frequent: sedation, rigidity, postural instability, falls and confusional state. The risk of mortality is doubled in patients treated with antipsychotics.
- Oculomotor disorders.
- Sleeping troubles. REM sleep (oneiric period of sleep) is characterized by muscle atony in healthy people. Patients with DLB develop REM sleep behavior disorders with violent shaking of the limbs during dream activity, excessive restlessness, persistent muscle tone, and absence of epileptiform electroencephalogram activity. The patient feels tired, but is usually unaware of this behavior. These disorders can be caused by cholinesterase inhibitors (eg Aricept, Exelon, Reminyl) prescribed for the treatment of cognitive disorders. Finally, a study has observed that these disorders can take years or even decades to dementia or Parkinson’s disease.
- Depression. Depression and apathy are common and may be related to the severity of movement disorders.
- Psychomotor restlessness.
- Delusions, often elaborate.
Diagnostic criteria
| Criteria | |
| Essential criteria | Gradual cognitive decline, Persistent memory impairment, not necessarily early stage, Deficit in attention and visual/spatial ability. |
| Important signs of the disease | Cognitive fluctuations, Recurrent visual hallucinations, Parkinsonian-like motor symptoms. |
| Exclusion criteria | Presence of cerebrovascular disease (stroke, transient ischemic) validated by cerebral imaging, Parkinsonian symptoms occurring only in the severe stage of dementia. |
These criteria make it possible to distinguish, with a small margin of error, a patient with MCI from a normal healthy subject (specificity* of the order of 80-100%). On the other hand, their sensitivity** is low and very variable (from 22 to 80%), suggesting that the prevalence of Lewy body dementia is underestimated.
It is estimated that an individual has an 80% risk of having MCI if they suffer from:
- of progressive dementia
- parkinsonian symptoms
- visual hallucinations
The medical community emphasizes fluctuations in alertness and attention.
Differential diagnosis
The doctor will rule out the diagnosis of LDB and will suspect another pathology depending on whether the symptoms are dominated by motor disorders of the Parkinsonian type, a cognitive deficit with loss of autonomy, or psychological and behavioral disorders. The conditions close to LDB on which the doctor will pay particular attention are the following:
- Alzheimer’s disease (AD). Disorders of episodic memory (recall of recent events) are earlier and more marked in AD than in DLB. Patients with Lewy body dementia, on the other hand, have a more impaired working memory, and more marked disturbances in attention, misidentification and delusional ideas. Visual hallucinations are observed later in AD. If the dementia appears more than a year before the onset of motor disorders, AD accompanied by a parkinsonian syndrome is preferred.
- Vascular dementia. The cognitive disorders observed in vascular dementia depend on the location of the cerebral infarction, and are sometimes accompanied by an apathetic state and motor disorders. Cerebrovascular lesions are uncommon in LDB.
- Parkinsonian type motor disorders. If the motor disorders precede the cognitive and behavioral disorders by more than a year, the presence of Parkinson’s disease or another form of parkinsonism is likely. Parkinson’s disease is likely if the patient suffers from tremors at rest and postural instability.
Other forms of parkinsonism can be considered such as:
- Progressive supranuclear palsy, differing from LDB by a more marked gait disorder and the absence of delusional ideas. The patient also has oculomotor disorders.
- Multiple system atrophy, which is very rarely accompanied by dementia. This parkinsonian syndrome is dominated by rigidity, akinesia (slow initiation of movements), and signs of dysautonomia (orthostatic hypotension, pupillary abnormalities, sphincter disorders).
- Corticobasal degeneration: apraxic disorders are more pronounced than in Lewy body dementia. The motor signs (akinesia, rigidity) are very asymmetrical.
- Creutzfeld-Jacob disease. Although the two diseases have common characteristics (abnormalities of the electroencephalogram, muscular contractions), the evolution of Creutzfeld-Jacob disease is generally fatal in less than a year. Analysis of the cerebrospinal fluid can help with the diagnosis, but only the autopsy can confirm it.
- Schizophrenia. Also called hallucinatory psychosis, this is preferred if the hallucinations are both visual and auditory (for example the patient hears voices telling him to harm himself), and are accompanied by delusions without cognitive decline or disorder engine.
- Metabolic dementia. Laboratory tests – blood tests, evaluation of thyroid function, blood levels of vitamin B12, brain imaging – will rule out or not vitamin B12 deficiency (often accompanied by psychosis), hypothyroidism (insufficient secretion of the thyroid resulting in psychomotor retardation, executive function disorders and psychosis). Cerebral imaging allows or not to eliminate the presence of lesions (lesions of the nuclei of the basal ganglia, of the thalamus and of the associative visual cortex) or cerebral anomalies (hydrocephalus, subdural hematoma) causing symptoms similar to those observed in Lewy body dementia. Imaging can also detect brain tumors affecting the lower temporal lobe and responsible for hallucinations and confusion. However, these tumors are rare.
Treatments of Lewy body dementia
Treatment of psychotic and behavioral disorders with antipsychotics is tricky because these patients are hypersensitive to this class of drugs.
This hypersensitivity aggravates the motor syndrome and induces states of acute agitation, leading to institutionalization.
Antipsychotics are the drug of choice, while limiting its dosage. Indeed, whether typical or atypical, they increase the risk:
- death and stroke in older people with dementia. The risk factors are: age over 65, existence of sedation, malnutrition, dehydration, lung disease or concomitant treatment with benzodiazepines.
- fall, this risk being already present in the Lewy body dementia.
They could also aggravate the cognitive deficit by promoting the appearance of lesions characteristic of Alzheimer’s disease (deposits of amyloid and neurofibrils).
Rivastigmine, a drug prescribed for cognitive disorders in Alzheimer’s disease, seems to reduce neuropsychiatric symptoms (eg delirium) in MCI and improve the quality of life of these patients.
Clinical case
An 80-year-old patient was hospitalized in a psychiatric unit for behavioral disorders (aggressiveness, confusional state) following treatment with an antipsychotic.
This person was previously diagnosed with dementia associated with a motor syndrome with:
- cognitive disorders: temporal, gnostic and practical spatial disorders;
- motor disorders: frozen face, voice and swallowing disorders, difficult walking.
He had been receiving rivastigmine and piribedil (a dopaminergic agent) for a year.
Despite the discontinuation of the antipsychotic treatment (replaced by an anxiolytic), the patient continued to exhibit agitation. Another anxiolytic was prescribed along with low-dose clozapine treatment. The gait disturbances were rapidly reduced, as well as his psychotic disturbances (hallucinations, agitation, aggressiveness).
